Why is Ice Less Dense Than Water
Ice is less denser than water because in ice the molecules arrange themselves in a rigid tetrahedral structure due to which cage like spaces remain in their bonding. If you put pressure on regular ice and give it time to rearrange the molecules will move into a new crystal lattice which results in the ice being more dense than water.
Together they are attached by a very strong bond called a covalent bond.

. The bond between the different water molecules is called a hydrogen bond this one is a little weaker. Ice floats because it is about 9 less dense than liquid water. This is what happens when enough thermal energy is present to break the rigid hydrogen bonds resulting in melting.
As you know two atoms of hydrogen and one atom of oxygen make water. Ice density waterWhy is the density of ice less than than the density of water why ice floats in water the density of ice and the density of water. Why is ice less dense than water when it freezes.
The heavier water displaces the lighter ice so ice floats to the top. When water freezes water molecules form a crystalline structure maintained by hydrogen bonding. Why is ice less dense than water intermolecular forces.
Ice is less dense than water because the orientation of hydrogen bonds causes molecules to push farther apart which lowers the density. The heavier water displaces the lighter ice so ice floats to the top. In other words ice takes up about 9 more space than water so a liter of ice weighs less than a liter water.
Ice is less dense than water because the orientation of hydrogen bonds causes molecules to. As it cools further and freezes into ice it actually becomes less dense. Cage like structure in ice and when the ice meltsfirst of all those bonds are brokenThusdue to than water molecules come closer and waters density increases and density of water is highest at 4cThats why water is more dense than ice.
Solid water or ice is less dense than liquid water. Why ice is floating on water. Yes which is precisely why ice frozen water floats in liquid water.
Thus the liquid form of water although. 11 Is Water Heavier When Its Frozen. Why is it important that ice is less dense than water.
This is because the hydrogen bonds in water are randomly orientated which means that the molecules are closer together. Therefore ice is less dense than water. The water molecules form crystalline lattice structures in ice where they are more spread out d.
As it cools further and freezes into ice it actually becomes less dense. In ice the hydrogen bonds hold the water molecules apart in an open lattice structure. Clearly once this crystaline structure is no longer forced into place by the rigid hydrogen bonding in ice it can collapse into itself resulting a greater density of water molecules.
Ice is less dense than water because the orientation of hydrogen bonds causes molecules to push farther apart which lowers the density. The water molecules are more closely packed together in ice than in water. Why is ice less dense than water.
The reason why ice is less dense than water is simple. Water is different because of hydrogen bonding. Why is ice not as dense than liquid water.
Ice is less dense than water because as water cools and becomes a solid freezes hydrogen bonds form between the water molecules. Ice floats because it is about 9 less dense than liquid water. Ice is less dense than water because the orientation of hydrogen bonds causes molecules to push farther apart which lowers the density.
But water molecules remain in linear bonding form. Solid water or ice is less dense than liquid water. As the volume of ice becomes greater it is less denser.
How bout this beastly answer. In the first ice crystal there are spaces between some of the molecules which is not there in the second crystal structure. In order for ice to be less dense than water the molecules in ice are more organized.
The water molecules in ice have less volume than in liquid ice. Yes some ice is denser than water. When water freezes water molecules form a crystalline structure maintained by hydrogen bonding.
In other words ice takes up about 9 more space than water so a liter of ice weighs less than a liter water. When water freezes water molecules form a crystalline structure maintained by hydrogen bonding. Martin Healey Studied Education Physical Sciences Upvoted by Kathryn Barker.
Solid water or ice is less dense than liquid water. This reduces the density of ice because the ice has more space between the molecules. The additional space created reduces the density of the water as it freezes making ice less dense than water.
Answer 1 of 5. Rather than the molecules packing more tightly together and creating a heavier density as water freezes the hydrogen bonds increase from a 34-molecule bond to a 4-molecule bond which. Water is considerably less dense than its solid counterpart ice and this unusual property is considered to be due to the hydrogen bonding in water.
The water molecules in ice have less mass than in liquid water b.
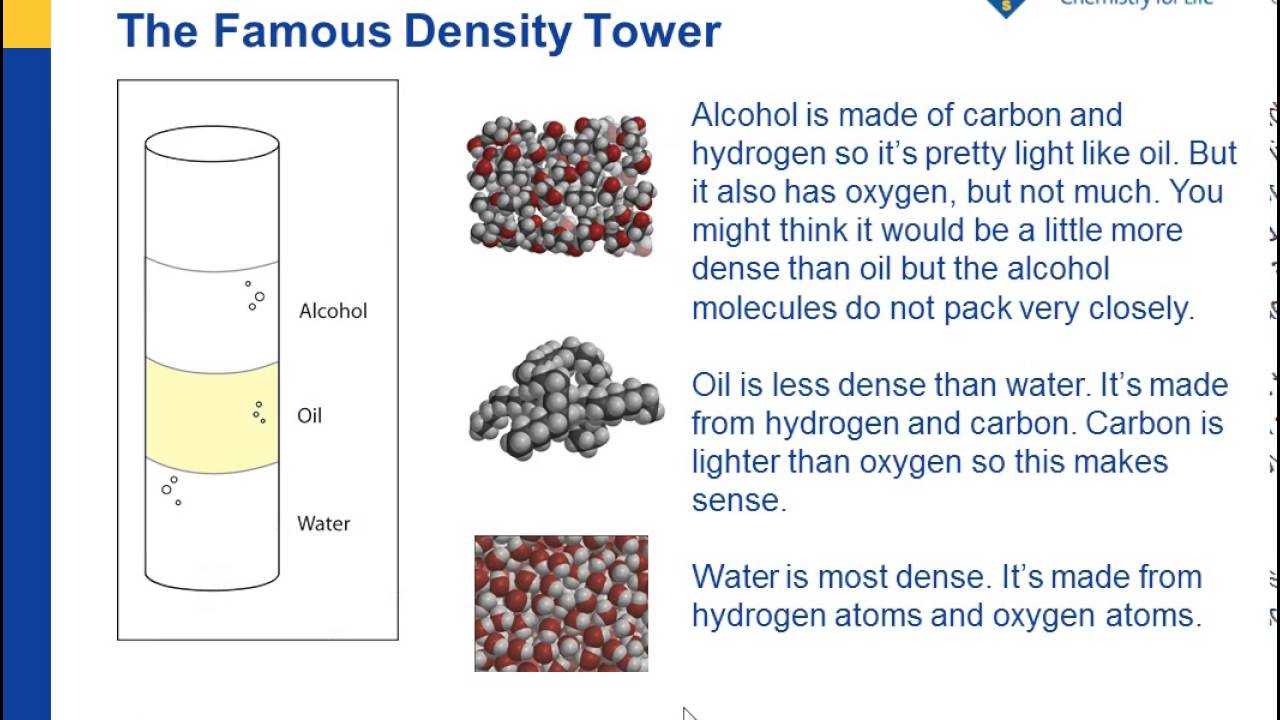
Density Sink And Float For Liquids Chapter 3 Density Middle School Chemistry Middle School Chemistry Teaching Chemistry Chemistry

Because Of Hydrogen Bonding Water Is Less Dense As A Solid Than It Is As A Liquid Consequently Ice Floats Language Chemistry Education

Ice Floats Because Solid Water Is Less Dense Than Liquid Water Arctic Landscape Landscape Outdoor

Ice Floats Ice Is Less Dense Than Liquid Water The Semi Crystalline Structure Forms Spaces Between The Molecules Chemistry Study Chemistry Life

Scientific Explanation Of Why Ice Floats Elementary Science Floating Hydrogen Bond

Pin By Debashis Panda On Chemistry Hydrogen Bond Water Bond Water Molecule

Comments
Post a Comment